LEADER IN THE NEUROTECH SPOTLIGHT: FRANK DESIERE, CEO COR-TEC NEURO , BOARD DIRECTOR & CHAIRMAN
ABOUT LIZ MOYLES – HEAD OF NEUROTECH, CRUXX, AUTHOR
Liz Moyles is a self-described frustrated neurosurgeon. A lengthy hospital stay in her teenage years left her with PTSD and a fear of hospitals and blood, rerouting her career path entirely at 18.
Medicine was the plan. Biochemistry became the reality. But life in a lab was not where she belonged. What never left her was her fascination with the brain and the people building in the space.
Today, as Head of Neurotech at Cruxx, she works with start-ups and scale-ups across the US, Europe and beyond to help them attract the talent and capital required to build.
Her aim is simple: to give CEOs and leadership teams their time back to operate at a strategic level, not get pulled into day-to-day execution.
Fundraising, clinical trials, and regulatory work already stretch teams thin. Hiring sits on top of all of it.
When it goes wrong or moves too slowly, the consequences are immediate. It can mean missing funding, delaying submissions, slipping milestones, or losing people through overload.
Liz leads global executive searches for VCs and neurotech companies, placing C-suite and board-level leaders and building out senior teams across the US, Europe and beyond.
Embedded in the neurotech ecosystem, she speaks regularly with founders, operators, KOLs and investors, giving her a clear view of where the field is heading and what it takes to succeed in a start-up. That perspective shapes how she approaches hiring.
In start-ups, every hire matters, and attracting the best is non-negotiable. But they are rarely looking. Busy building elsewhere and often unaware of what you’re doing.
Finding them is one challenge. Getting them to engage is another.
But while you and your hiring managers are trying to find these people, the clock is running. FDA submissions, fundraising rounds, clinical trials.
Everything that cannot wait is …………waiting.
Reach out to Liz. That person is out there. She knows the space and where to find the people.
They just don’t know you exist. Yet!
ONE SMALL GERMAN BCI COMPANY STEALS THE LIMELIGHT !
I interviewed Frank Desiere, CEO of Cor-Tec, a few months ago for my Leader in The Spotlight feature.
Given the announcement 29 April 2026, I thought it would be timely to reshare Frank’s interview so we can read it given the subsequent events we are now aware of.
At the time Frank and I recorded the interview, the story of Cor-Tec’s achievements was already remarkable.
Matt Kidd, a Seattle mechanic and a two-time stroke patient had been told his recovery had plateaued and was classified by practitioners as ‘a dead end’.
Within 6 weeksof being implanted with Cor-Tec’s Brain Interchange ™, he regained more motor function than in the previous year of conventional therapy.
The gains held and continued to improve even after stimulation stopped. He got to the point where he could lift his dog, Bentley, with his arm, open his shower curtain and much more.
Since that conversation, with Frank in February, Cor-Tec’s April announcements have made this one of the most significant months in the history of implantable BCIs.
April 8: FDA Breakthrough Device Designation, the first BCI worldwide designated specifically for stroke motor rehabilitation.
April 23: FDA TAP Program acceptance and a third patient successfully implanted at Harborview Medical Center in Seattle.
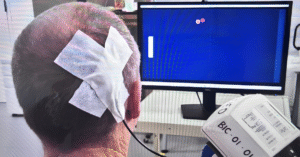
April 29: Mind control – The same implant that helped Matt recover motor function enabled him to control a computer using only his thoughts.
No New Hardware. No Additional Surgery. Same Device.
To our knowledge, Brain Interchange ™ is now; the first and only fully implanted, wireless BCI worldwide to deliver both therapeutic stimulation and thought-based computer control in the same patient using identical hardware.
THREE-TWO-ONE. 2026 – BLASTOFF FOR COR-TEC!
Three Milestones In Three Weeks. Two Clinical Breakthroughs. One Device.
Every other major BCI programme today, Neuralink, Synchron, Blackrock Neuro, Precision Neuroscience, is built around helping people live with a disability.
But ultimately the disability remains and the therapeutic intervention, if there is one, lies separate and downstream.
Cor-Tec is aiming to reduce the disability itself.
That distinction matters as Brain Interchange ™ is targeting 4 indications on the same implant:
-Stroke rehabilitation
-Epilepsy with Mayo Clinic
-BCI for paralysis and communication and
-Treatment-resistant depression, (TRD) planned with University Hospital Freiburg.
The implications of this are phenomenal and it appears that Cor-Tec, one small, German, relatively modestly funded ($28M) MedTech company has forged way ahead of all its more richly funded US and Chinese counterparts!
What follows now is the original post:
In early February Cor-Tec announced a further major milestone. The successful second implantation at Harborview Medical Center in Seattle. The procedure went smoothly, and the new participant is recovering as expected.
I have known Frank Desiere, CEO and Martin Schuettler, Co-Founder & CTO for quite some time now, so I jumped on a call with them to hear their account of what had happened. It was truly emotional to hear the excitement, mixed with pride, and a sense of deep compassion for this latest participant whose life is set to change for the best.
Before I dive into the interview with Frank, I thought I’d share a brief intro.

THE PEOPLE BEHIND COR-TEC & THEIR VALUES
At the heart of Cor-Tec’s neurotechnology journey are three very different people united by a shared conviction: that the brain’s capacity to recover and adapt is greater than we’ve long assumed, and that technology can help unlock that potential for patients.
Co-founder and CTOMartin Schuettler laid the technical foundation. An engineer deeply immersed in neural interfaces and implantable systems, he helped shape Cor-Tec’s core capability building devices that can safely and meaningfully interact with the brain.
CEO Dr. Frank Desiere has a background spanning life sciences, business, and global medtech leadership, he is steering Cor-Tec from pioneering research toward scalable clinical impact. His leadership is shaping Cor-Tec’s transition from pioneering engineering to scalable clinical impact.
Both share the same beliefs and anchor Cor-Tec’s direction in clinical reality, ensuring the technology is never just about hardware, but about patients.
They believe the brain can be an active partner in recovery, and that technology should be built to serve people, not just advance capabilities.
A DEAD END!
Doctors described Matt Kidd, a 53-year-old mechanic whose life changed drastically after a second stroke as having reached a ‘dead end’.
Twelve months of rehabilitation and changes had plateaued.
Matt had his first stroke in 2012, after which he relearnt how to walk and talk.
His second stroke hit on Memorial Day 2021 after he had spent the holiday at the Packwood Flea Market.
At a family barbeque that evening, Matt told everyone he was going home because he didn’t feel quite right.
He drove himself to Harborview. He was admitted, given a shot, and spent the night at the hospital.
“When I woke up, I couldn’t move my left arm and my left leg,” he said.
Matt was transferred immediately to rehab, where he remained for a month. But the focus of his physical therapy was mostly on getting him up and walking, his longtime partner Tessa Kidd said.
Despite intense physiotherapy and rehabilitation, his recovery had stalled. When the study began, Matt could barely hold a broom handle out in front of him with both hands.
Matt Kidd was the first participant to receive Cor-Tec’s fully implantable Brain Interchange ™ system in summer 2025.
Paired with rehabilitation, he regained meaningful function in his arm and hand – and was able to hold a cup, unscrew a nut and close the shower curtain just 6 weeks with the Brain Interchange™ system + rehab.
The doctors were amazed.
Matt was overwhelmed with emotions and gratitude. In time he was able to lift Bentley, his dog onto his lap.

Liz: Frank- can we start by asking what Cor-Tec does and the problem it is built to solve?
Frank: Yes of course.
Cor-Tecis a German neurotechnology company founded in 2010 in Freiburg.
We do two things.
First, we are a development and manufacturing partner for advanced components and active implantable medical devices – electrodes, hermetic packages, and complete implantable systems. That business is growing at more than 20% annually and serves a real need in the industry.
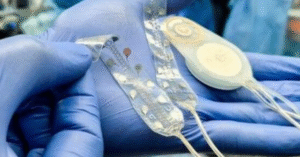
Second, and this is the heart of what drives us, we have developed the Brain Interchange™ – a fully implantable, wireless brain-computer interface, (BCI), platform.
The problem we set out to solve is fundamental: millions of people with neurological conditions such as stroke, epilepsy, depression, movement disorders, reach a ceiling with existing therapies.
Physical therapy and medication do important work, but for many patients, particularly in stroke rehabilitation, they are simply not enough.
Over 80% of stroke patients experience upper-limb impairment acutely, and more than 40% remain impaired chronically.
In the US and Europe alone, 1.7 million people have a stroke each year.
Only about 10% recover fully. The rest live with permanent limitations that affect their independence, their livelihoods, and their families.
Our platform addresses this by creating a direct, bidirectional dialogue with the brain. It continuously records neural activity and delivers targeted electrical stimulation in real time – a closed-loop system.
The idea is to enhance neuroplasticity, helping the brain rewire itself and relearn lost functions.
We are the first company in Europe, and the first German company, to bring a fully implantable bi-directional BCI system into FDA-approved clinical evaluation in the United States.
SUPERCHARGING NEURAL ACTIVITY AT THE BRAIN LEVEL
Liz: I am so proud of you all. Tell me – how does this compare to the current standard of care?
Frank: Today, the standard of care for stroke rehabilitation is essentially physical therapy and occupational therapy- task-specific training, constraint-induced movement therapy, and in some cases functional electrical stimulation of muscles or botulinum toxin for spasticity.
These approaches are valuable, but they have real limitations.
The recovery curve is steep in the first 3 months and then largely plateaus. After 6 months, most clinicians will tell you the window for meaningful gains has narrowed significantly. The evidence also shows that rehabilitation sessions are often too few and too short, and far too little time is dedicated specifically to the upper limb.
What we offer is fundamentally different.
Our system works at the level of the brain itself. Rather than stimulating muscles or relying solely on voluntary effort, we place electrodes directly on the cortical surface and use real-time, closed-loop stimulation to induce neurons to fire together during rehabilitation.
The timing is critical – you want to activate the neurons precisely when the brain is attempting a movement, so the two brain areas train together.
This is not about replacing physical therapy; it is about supercharging it at the neural level.
The key differentiator of Brain Interchange ™ is its bidirectional, closed-loop capability.
It both reads and writes – it records brain signals and responds with adaptive stimulation in real time.
Most existing neuromodulation devices are either read-only or write-only.
Our system does both simultaneously, and it does so wirelessly with no cables and no physical links through the skin.
That is a meaningful step forward.
Liz: So, it’s not meant to replace rehab, but to amplify it at the level of the brain. The obvious next question is whether that translates into meaningful change for patients. What’s the moment that has made you most proud so far?
Frank: Without question, let’s discuss the recovery of our first study participant, Matt Kidd.
Matt is a 53-year-old mechanic from the Seattle area who suffered multiple strokes, including a severe event in 2021 that left him with significant impairment on his left side. He had undergone more than a year of conventional physical therapy with limited progress.
He had reached what his doctors described as a dead end.
After just 6 weeks of rehabilitation combined with stimulation from our implanted device, Matt regained more upper-limb function than he had during the entire preceding year of therapy.
He could hold a cup, unscrew a nut, close the shower curtain – daily activities that most of us take for granted but that represent a profound change in independence and dignity for a stroke survivor.

He could lift the broom handle he could barely hold before the implant, over his head with 8lbs weights on either end.
Tessa, his wife described seeing the side-by-side video comparisons as “breath-taking.” And what is particularly encouraging is that even after the stimulation was turned off, the gains persisted, which suggests genuine neural rewiring, neuroplasticity, had occurred.
Matt’s progress was so significant that, as his physicians noted, if he applied for the study today, he would no longer qualify- his function has improved beyond the inclusion criteria.
That is the kind of result that keeps us going.
FOUR CONDITIONS – ONE IMPLANT
Beyond stroke, I am also very proud that our platform is already generating scientific results in epilepsy.
We have a registered clinical trial at Mayo Clinic – NCT05439655- led by Professor Nuri Ince and Professor Gregory Worrell, investigating the use of Brain Interchange ™ for closed-loop seizure control in drug-resistant epilepsy.
What makes this trial particularly compelling is the approach: rather than waiting for a seizure to begin before delivering stimulation, as current responsive neurostimulation devices do, the Brain Interchange™ system monitors stereotyped high-frequency oscillations – early biomarkers of seizure onset – and delivers targeted stimulation to prevent seizures from occurring in the first place.
The team has already validated that our system captures these HFO signals with fidelity comparable to established clinical amplifiers, published in Nature Scientific Reports.
If the acute-setting results continue to be positive, the next step is a chronic ambulatory clinical trial with the fully implantable system.
And in parallel, we are working with Professor Nick Ramsey’s group at UMC Utrecht as part of the INTENSE consortium, developing next-generation implantable BCI technology for people with severe paralysis.
Cor-Tec is providing customised high-density electrodes and implantable electronics for that programme.
So, the clinical footprint is expanding ;
-Stroke
-Epilepsy
-Paralysis and we plan to study
-Treatment resistant depression also.
Each collaboration strengthens the evidence base for our platform.
THE SECOND SUCCESSFUL IMPLANTATION!
And then today, 10 February 2026, we announced the successful second implantation at Harborview Medical Center in Seattle.
The procedure went smoothly, and the new participant is recovering as expected. For our clinical partners at the University of Washington and UCLA, and for our team in Freiburg, this is validation that we are on the right path.

CHALLENGING ACCEPTED NORMS
Liz: Matt’s story is striking, not just because of the gains, but because of how far out he was from his stroke. It challenges some long-held beliefs in neurology. What assumptions do you think are being overturned most right now in how we treat neurological disease?
Frank: Three assumptions are being fundamentally overturned.
FIRST: The Brain Cannot Recover Beyond A Certain Point.
For decades, the prevailing view was that after a relatively short window post-injury, neurological recovery plateaus and that is essentially the end of the story. Our data, and data from other groups, is challenging that directly.
Matt Kidd was years out from his stroke. The brain retains a capacity for plasticity that we have systematically underestimated.
SECOND: Pharmacological Intervention Is The Primary Tool For Neurological Conditions.
We are seeing the beginning of a paradigm shift from chemical pharmacology to bioelectronic medicine- from pills to pulses, if you will.
Devices that interact directly with neural circuits are starting to demonstrate efficacy in conditions from epilepsy to depression to movement disorders. This is not about replacing drugs entirely, but about opening an entirely new therapeutic modality.
THIRD: The Brain Is A Black Box.
This is perhaps the most profound. The assumption that the brain is a black boxwe can scan but not truly interact with.
BCIs are changing that.
We can now have a real-time conversation with the brain- listen to what it is doing and respond intelligently. That capability opens a frontier for adaptive, personalised therapy that was simply not possible before.
Liz: That shift from “the brain can’t change” to “the brain can be trained” is a big deal.
But some conditions move faster than others when it comes to changing clinical practice. Which neurological areas do you think are closest to seeing real shifts in standard of care?
Frank: Epilepsy is arguably the furthest along.
Closed-loop neuromodulation for drug-resistant epilepsy is already an approved therapy – NeuroPace’s RNS System, for example, has demonstrated meaningful seizure reduction across thousands of patients.
But the current generation of devices reacts to seizures after they start.
The next step here is seizure prevention. That is precisely what our registered clinical trial at Mayo Clinic is investigating – Professors Nuri Ince and Gregory Worrell are using the Brain Interchange™ to detect stereotyped high-frequency oscillations, early biomarkers of seizure onset, and deliver targeted stimulation before a seizure can develop.
Stroke rehabilitation is, I believe, the next major frontier.
What we are seeing in our study, and what the broader research community is demonstrating, is that targeted brain stimulation combined with rehabilitation can push beyond the traditional recovery ceiling.
Vagus nerve stimulation paired with rehabilitation has also recently gained traction. As more clinical data accumulates, I expect stroke to be a condition where BCI-assisted therapy becomes part of the clinical pathway within this decade.
Treatment-resistant depression is another area to watch closely.
Flow Neuroscience recently received FDA approval for at-home neuromodulation for depression, and several groups are exploring implantable approaches for severe cases.
And for patients with severe paralysis from conditions like ALS, BCI-enabled communication is progressing rapidly. At UMC Utrecht, Professor Nick Ramsey’s team- with whom we are partnering in the Dutch INTENSE consortium- previously demonstrated the world’s first fully implanted home-use BCI for a locked-in ALS patient.
We are now working together to develop the next generation of that technology, with plans to trial the system in paralysis patients in Utrecht and in Graz.
Cor-Tec is providing the customised high-resolution electrodes and implantable electronics. This is an area where the unmet need is existential- these patients are trapped in their own bodies- and BCI technology is the most promising path to restoring communication.
Our platform is designed as a versatile tool. While stroke is our initial clinical focus, we are already in discussions with research groups on epilepsy, movement disorders, spinal cord injury, and psychiatric conditions.
The same closed-loop architecture applies across indications.

Liz: What trends or signals are you watching most closely in MedTech, particularly in the neuro space, this year?
Frank: There are several.
FIRST: The Convergence of AI And Neural Interfaces.
The ability to decode brain signals in real time is being transformed by machine learning. The systems that will win are those that can combine high-fidelity neural recording with intelligent, adaptive algorithms. This is precisely where our closed-loop platform sits, and it is where the field is headed.
SECOND: The Race For Better Form Factors And Less Invasive Approaches.
There is significant innovation happening in electrode design – from endovascular approaches like Synchron’s Stentrode to high-density micro-arrays. We have chosen a semi-invasive, epidural approach that balances signal quality with safety, and I think the field will increasingly recognise that there is no one-size-fits-all solution.
Different indications will require different levels of invasiveness.
THIRD: THE COMPETITIVE LANDSCAPE IS INTENSIFYING
That is a good thing. Major strategic players are entering – Medtronic’s partnership with Precision Neuroscience is a notable signal. Capital is flowing in.
The global BCI market was valued at roughly $2.8 billion in 2024 and is projected to exceed $11 billion by 2033. That growth attracts talent, investment, and clinical interest.
FOURTH: THE REGULATORY, FUNDING & GEO-POLITICAL ENVIRONMENT.
The FDA has granted Breakthrough Device Designation to several BCI companies, which accelerates development.
Funding uncertainties in the US, particularly around NIH, are a factor that the entire research community is navigating.
In Europe, the MDR framework presents both challenges and opportunities.
And frankly, I am also monitoring the geopolitical dimension – Chinese competition in BCI is accelerating.
Liz: It’s interesting that you’re talking about stroke, epilepsy, depression, paralysis. That’s a huge clinical landscape. If we fast-forward to the end of 2026, what would success look like for you and your team? And what excites you most about what comes next?
Frank: By end of 2026, success means several things:
Clinically, we want to have enrolled and treated additional patients in the safety phase of the ongoing study at the University of Washington and to have compelling, peer-reviewed data demonstrating the safety and therapeutic potential of our platform.
We have two successful implantations now; the study will enrol further participants, and each one gives us richer neural and behavioural data.
Commercially, we are actively evaluating strategic options to fuel the next stage of our development-– that includes further fundraising, but also strategic partnerships and collaborations with larger industry players.
We have received takeover indications as well, which speaks to the value the market sees in our platform and clinical assets.
Our priority is to find the path that best accelerates patient access to this technology while preserving the innovation culture that has brought us this far.
Strategically, we want to deepen our clinical collaborations across multiple indications.
Beyond advancing our stroke programme with the University of Washington and UCLA, we aim to progress our epilepsy trial at Mayo Clinic with Professors Ince and Worrell – the acute-setting work on seizure prevention using stereotyped HFO detection is showing promise, and positive results would set the stage for a chronic ambulatory trial with the fully implantable Brain Interchange™ system.
With UMC Utrecht, we are working toward the planned INTENSE clinical trial implanting our system in severe paralysed patients – that would represent a major milestone in demonstrating the platform’s versatility across neurological conditions.
We are also exploring how to accelerate the path toward pivotal trials and, ultimately, regulatory approval.

What excites me most? It is the moment we are in.
For 15 years, Cor-Tec has been building the engineering and science to make this possible.
Now, for the first time, we are seeing real patients experience real recovery with our technology.
Matt Kidd’s story is not just a data point – it is a human being getting his independence back.
The fact that the gains persist after stimulation is turned off tells us something profound about what is possible.
We are at the beginning of a new class of neurotherapeutic solutions, and I am deeply motivated to see where this leads – for stroke, and ultimately for the millions of patients worldwide living with neurological conditions who deserve better options than what exists today.
Liz: Thanks so much Frank, Martin and team and well done.
REFLECTIONS – FEBRUARY 2026
What stayed with me most after speaking with Frank wasn’t the technology. It was the humanity behind it.
When a stroke survivor goes from “there’s nothing more we can do” to holding a cup, closing a shower curtain, and regaining dignity in daily life, it forces a bigger question: how many people are living below their potential simply because our tools have not caught up yet?
Cor-Tec’s second implantation is another important step. Now the work becomes proving this can be repeated, scaled, and ultimately built into a clinical pathway.
FAST FORWARD TO MAY 2026.
Revisiting my February conversation with Frank, and reading CorTec’s April 29 announcement, one thing stands out:
The speed at which BCI and neurotechnology are advancing.
In February, the focus was clear. Restore motor function post-stroke.
Now, with the same fully implanted, wireless system, we are seeing something more: The ability to decode intent and translate it into real-time control of an external system.
No additional surgery. No new hardware. That matters.
It demonstrates something deeper about the platform. The boundary between therapeutic recovery and interaction with the external world is thinner than many expected.
THIS IS WHERE IT BECOMES BOTH EXCITING AND UNCOMFORTABLE.
We are not just improving outcomes within a defined clinical use case. We are starting to extend capability into adjacent domains, using the same underlying system.
That suggests we are beginning to move beyond established norms and, slowly, into what has historically been treated as a black box.
It’s difficult not to be excited by that. At the same time, for the Neurotech Community and the World, it raises questions we need to take seriously:
-Ethics – and whose definition of ethics applies here? Do we have accepted agreed universal ethics?
-Do we need, a shared global standard?
-How does regulation keep pace with capability?
-Where is the line, and who decides when it is crossed?
Because this will not slow down. And if the technology is moving this quickly, the frameworks around it will need to move just as fast.
 Understanding The Brain – The Ultimate Frontier
Understanding The Brain – The Ultimate Frontier ! – by a frustrated Neurosurgeon
! – by a frustrated Neurosurgeon




