LEADER IN THE NEUROTECH SPOTLIGHT: DAVID VEINO, PRESIDENT & CEO, NEUROS MEDICAL

NeuroTech often focuses on expanding markets: new indications, larger patient populations, bigger total addressable markets.
David Veino, CEO, is building a company in a space where, paradoxically, the best long-term outcome would be that the market shrinks.
That would be my wish, but more importantly it is David’s wish too.
Neuros Medical is developing a therapy for chronic post-amputation pain, a condition affecting a large portion of amputees who often cycle through systemic drugs, opioids, repeat procedures, and persistent pain that can profoundly affect quality of life.
I have to say, I have interviewed many CEOs, and David is a rare breed.
He genuinely never stops thinking about the individuals using Altius, or those who don’t have it yet, and the responsibility he feels for the people, who in spite of having previously been let down by others, and the system, put their trust in him and Neuros.
It weighs heavily on his mind but for all the right reasons. And it’s a weight he would be reluctant to lose to give up.
On a dull Monday morning, he brightened up my day with his passion, his smile and his story.
In this conversation, David reflects on how he came into the field, why amputee pain remains one of the most underserved areas in MedTech, and why establishing a true standard of care matters more to him than simply building another neuromodulation company.
TAKE CARE OF YOUR MUM
Liz: David, I’m curious. When you were a wee lad, kicking a ball around the field, did you dream, of becoming involved in MedTech?
David: Not quite. I knew I wanted to be in the Medical world but in a different capacity. And at 22, my entire path changed. I had been accepted to medical school. I wanted to be a trauma surgeon.
Then my dad passed away on the day I graduated from college.
On his deathbed, his direction to me was simple: do whatever you can to take care of your mom.
That changed everything.
I had to make decisions through a different lens. I knew I still wanted to make a difference to the health of individuals.
I stumbled into the strange world of medical devices, which I never thought I would be a part of, but the mission pulled me in immediately.
And the longer I have been in it, the more I realize this field is really about people – not patients.
It’s about the individual, understanding their background, their hopes, what makes them who they are, and why they do what they do.
We, in general do not take time out to speak to people really any more. We are texting, slacking, and generally communicating in other ways rather than a real conversation.
I think that is also true in the medical world. We do not take enough time to see the human being behind the ‘patients’.
That’s what I see too often – not always. That’s what my team see.
That is what many of our ‘patients’ have said. Time and time again.
It makes me sad.
Liz: When I went through breast cancer, chemotherapy and radiotherapy, the only 4 people that treated me as a human were the phlebotomist, the two receptionists and the breast surgeon. I remember him – he looked at me and said “Liz, you’re young, strong and very fit but I am so sorry about what I am going to put you through. It’s going to be hard.”
It was the only time I heard anything like that. When I met the oncologists and the majority of others- I felt like a number. That they had forgotten that behind the cancer, behind the patient was a human. Maybe it’s a self defence mechanism.
But tell me about your career and experience before Neuros.
WHAT HAPPENS AFTER AMPUTATION

David: I’ve spent about thirty years in medical device. Most of that time was working with physicians in areas like cardiovascular, peripheral vascular disease, spine, and interventional pain.
Earlier in my career I had the opportunity to work at companies like Stryker and a number of other organizations focused on developing technologies that could truly change health outcomes.
One of the areas I spent a lot of time in was peripheral vascular intervention, particularly limb salvage. That meant working with vascular surgeons treating individuals with severe peripheral artery disease who were at risk of losing their limbs.
That experience is actually what led me toward the amputee population. We would save limbs when we could, but when we had to amputate, I started asking what happened to those patients afterwards.
Sometimes, very rarely, we would see them come back into the cath lab for contralateral limb revascularization or something else, but not as often as I expected.
I couldn’t help but wonder, what happens after amputation. What is the patient journey? What is the after care?
What is their life like after amputation?
It weighed on me.
BETRAYED, FORGOTTEN, INVISIBLE, IN AGONY
No one knew.
I started talking – asking about the patients. Thinking about the human being inside the patient, back, in effect to the point I made when we started this discussion Liz.
What I learned was not great. I began reaching out and speaking to individuals and associations.
I was told time and again that many felt failed, betrayed, forgotten.
They talked about a loss of dignity, pride, independence. About feeling ‘a burden’.
They felt the systems had let them down- the medical system, the psychiatric / psychological support system, the healthcare system, their employers, sometimes even their friends and families.
And they all talked about the agonising, relentless pain.
Phantom limb and residual limb pain.
Many are living with chronic pain.
That pain becomes compounded with depression, anxiety, PTSD, and all the things that follow. It becomes this downward spiral where people lose independence and quality of life.
Liz: I have known people in that situation, and I have seen them trying to get their life together. Some at a very young age, after a life changing accident. But I have never felt it my place to ask about the pain. When someone develops chronic post-amputation pain, what options do they have?
David: The first-place individuals are usually directed to is systemic drugs.
If those, don’t work or don’t work sufficiently, they may be moved onto opioids or gabapentinoids. Those bring their own challenges around dependency and drug interactions.
The individuals told me that in many cases their lives centered around watching a clock ticking. Waiting for their next pill.
Other options for physicians include performing a nerve block with lidocaine. It blocks the sodium channels that carry the signal. But you cannot repeat this indefinitely because it becomes toxic to the nerve.
A further option is the surgical removement of a painful neuroma. But neuromas grow back. Histologically you will see them start regrowing within ninety days, and sometimes they come back even more painful.

Then there are other peripheral nerve stimulators. Many of those technologies add additional signal to the central nervous system.
If the problem with chronic post-amputation pain is already a flood of signals going to the brain, the last thing you want to do is flood the system with more signal.
That is why treating the post amputation pain has been such a difficult condition to treat.
Many of these individuals feel they have no options left. There are no real avenues to escape the excrutiating pain or the consequences of the treatments they have already endured.
They desperately need something that can relieve their pain and restore a life that matters to them. We can’t give them back their lives as they were before- because that has changed.
For some, the despair becomes overwhelming and suicide feels like the only escape.
My team and I want to try to stop that ever happening again.
These are individuals who have lived through something most of us could never truly understand while having intact limbs. And many have lost trust too.
In our organization we always say: Relieve Pain and Restore Life.
To me that matters.
A CRAZY IDEA
Liz: I can’t tell you how horrid and sad that is to hear. Thanks for sharing David. Thank you. Thanks also for explaining the alternative treatments. So tell me more about Neuros and Altius.
David: If neuromodulation is finally attempted with an individual, most of the neuromodulation technologies are based on the gate control theory of pain. The idea is that if you flood the nervous system with stimulus, this reaches the brain faster, and you can alter the perception of pain.
In effect the same level of pain is there, but it is felt less acutely.
Our device, Altius, works differently.
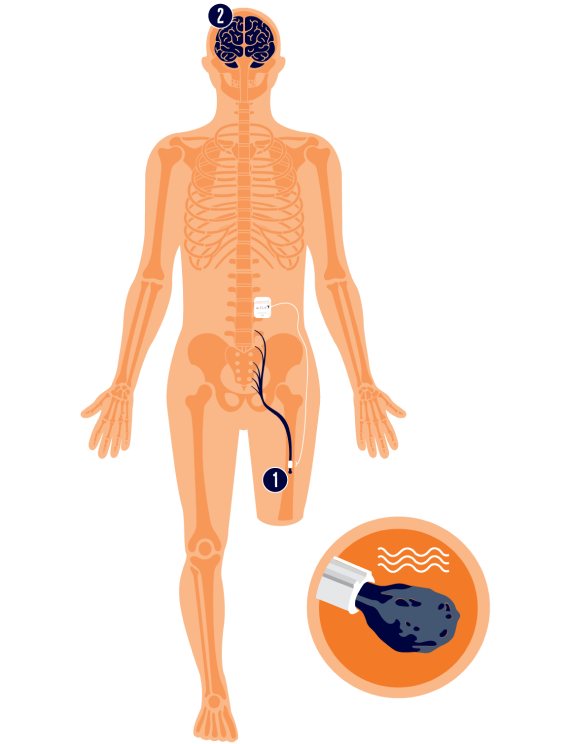
In fact when people first heard about it, they thought the idea sounded crazy because our approach is to block both sensory and motor signals.
Liz: Now you say that I can see how it would work with amputation, but that is an incredible idea. Where, and who came up with it ?
David: The concept actually came out of Case Western Reserve University in Cleveland.
A team had done some early benchtop and animal work showing that high-frequency, high-amplitude energy could block nerve signaling. But they had not turned it into a product.
In essence it was an idea, searching for a solution.
Our founder came across that IP while working as a CEO-in-residence and believed it could become a real technology.
But when you apply it to a severed nerve in an amputee, the physiology makes it a perfect fit.
From there the work was turning that concept into a clinical device. Understanding the frequencies required, how to deliver the energy safely, and how to design the cuff electrode that wraps around the nerve.
Liz: So stepping back a moment, David, just before you explain, can I ask – how big is this system ? how visible is it because I imagine this population may already feel ‘self-conscious’ about body image.
David: You’re right – and that’s why so much thought has gone into the aesthetics and size. We involve ‘patients’ from the start. We need to know what really matters to them, rather than assume.
Often what we may think matters, is not really the case, and other less obvious factors, are the more important ones.
From the outside, most people would never notice the device at all. The system is fully implanted. A cuff electrode is placed around the nerve at the site of the amputation. The lead is then placed underneath the skin and connected to a small implantable pulse generator.
That pulse generator is typically placed in the abdominal area just under the skin- so that it is quite disguised.
The individual then has a controller to manage the therapy for themselves. When they feel pain starting, they activate the device using the controller. The therapy Altius delivers lasts for just thirty minutes and then automatically shuts off.
That 30 minutes means the majority of individuals get 7-9 hours of pain relief from a single therapy session.
Liz: Does blocking the nerve affect the ability to move?
David: While the therapy is running it blocks both sensory and motor signals, which is why we ask the individuals remain seated during the thirty-minute therapy session- but after that, they can do what they want.
Liz: Something that really struck me reading your profile, is I assumed you had always been in neuromodulation. I never thought about it when we met and I never asked. But this was your first foray into that space.
I recruit extensively at your level and one of the biggest constraints Boards and CEOs place on a search is previous domain experience – “they must have previous experience”.
How and who gave you the opportunity to step into this space?
David: The previous CEO of Neuros, Tom Wilder, was the person who brought me into the company.
Tom had a long career in medical devices, including time at Medtronic and building other businesses. He had been working with the investors who had backed Neuros, including US Venture Partners.
He approached me and said he was looking for someone who could help build the company and take on a broader operational role.
At the time, I had a background in vascular, spine, and pain markets, but I had not worked specifically in neuromodulation.
What I did have was experience building businesses and working closely with physicians in areas where the technologies really mattered to the end users- the individuals.
Tom gave me the opportunity to come in and take responsibility for areas like clinical operations, R&D, regulatory, and quality. That allowed me to learn the technology and help shape how the company would grow.
I have a lot to thank Tom for. He gave me the chance to expand into areas of the business that I had always wanted to understand more deeply.
A SIMPLE QUESTION- THE PIVOT
Liz: It must have been a lot to take in all at once. What were your immediate observations?
David: It was a lot to take in. When I arrived, the technology was incredible, but the company was struggling to enrol the trial. I noticed, the investigators were mainly interventional pain specialists and neurosurgeons. The challenge is that those physicians do not typically see a lot of amputees.
Because of my background in peripheral vascular disease and limb salvage, I had spent years working with vascular surgeons. They are the physicians who perform the majority of amputations and continue to manage these ‘patients’ afterwards.
So I stepped back and asked a simple question: “who typically sees these ‘patients’?
That led us to pivot the trial toward vascular surgeons as investigators. Once we made that change, enrolment accelerated.
Within about eighteen months, during the height of COVID, we completed enrolment ahead of schedule. That was one of the most remarkable things I have seen in my career.
The individuals we contacted to take part in the trial to test this technology, FOUGHT through all the challenges and restrictions in place during COVID to get to those sites because they were desperate for a treatment option.
Liz: How quickly was it before the individuals who applied to be part of the trial found out if they were eligible and accepted, and whether they would be a responder?
David: This is another important point. Often individuals have to wait for weeks or longer to learn if they have been successful in their application to be part of a trial. It’s hard.
Instead of doing a two-week trial implant, we use a lidocaine injection under ultrasound directly on the nerve.
Because lidocaine blocks the same sodium channels our therapy targets, it becomes a direct comparator.
Within about twenty minutes we know whether the individual is a responder.
If their pain drops by 50% with that injection, they have a high likelihood of successful results with the therapy.
Liz: That is incredible, because even that wait, the 2 weeks, can seem like a lifetime, and if you have raised hopes that you may be a responder, to have to wait 2 weeks and then discover you are not eligible, must feel like yet another cruel blow.
What did the results of that pivotal trial ultimately demonstrate?
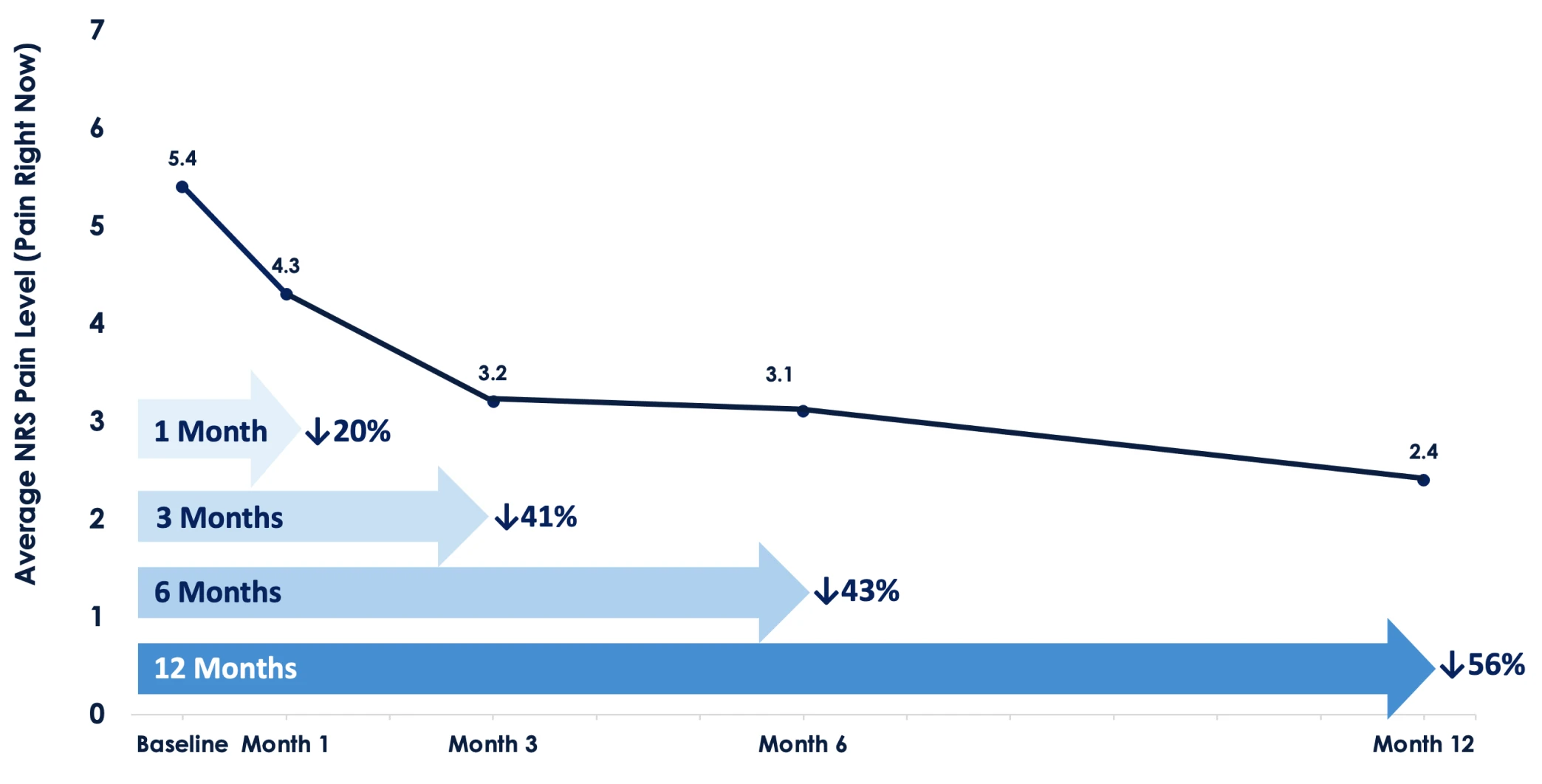
David: It was a randomized, double-blind study against an active sham control. The hardest type of trial you can run.
We saw a 45% improvement in quality of life, including improvements in walking, relationships, and ability to work.
We also saw an 81% reduction in opioid use overall and 35% of the individuals actually stopped taking opioids altogether.
As I said, using our system, the individuals typically receive around 7-9 hours of pain relief from a thirty-minute therapy session.
Over time we also saw reductions in both acute pain and chronic pain levels.
A LEAP OF FAITH
Liz: When you first started speaking with investors about Neuros, how did they react to the idea of building a therapy specifically for amputees?
David: If you look at the numbers, there are more than 2,000,000 lower limb amputees in the United States today.
Up to 80% of them will experience chronic post-amputation pain at some point. And there are roughly 150,000 new major lower limb amputations every year.
But once you account for comorbidities, mortality, and which individuals are actually good surgical candidates, the true addressable population becomes smaller.
You are probably looking at something closer to about 1,000,000 individuals in the United States. So yes, there is a real ‘patient’ population.
But the challenge was that nobody had ever really studied amputees in this way before. There had never been a dedicated technology developed specifically for them.
And, if there has never been a solution before, some people start to question whether the problem really exists at the scale you are describing.
That meant many investors struggled to understand the market opportunity.
Liz: So you had to find investors who were comfortable backing a very novel technology whilst also asking them to be there with you whilst you created a and new market.
Wow. That was a big ask David.
David: It was. We did our homework. Some of the early investors had experience backing novel technologies before. Those early investors were incredibly important.
They understood that sometimes a new technology creates a market that did not exist before.
When you are developing something that is completely new, many investors struggle to understand the opportunity because the market has not really been defined yet.
So, the people who step in at that stage are taking a real leap of faith. They believe in the technology and the mission before the clinical data or the market is fully established.
Liz: Can you tell me about investors?
David: Over the years the company has raised approximately $150M to develop and bring this technology to market.
The most recent financing was a $56M Series D, led by EQT Life Sciences, with continued participation from investors who have supported the company over time, including US Venture Partners, Aperture Venture Partners, Osage University Partners, and others who understood that sometimes a technology creates a market rather than entering one. Boston Scientific was and remains involved as a strategic investor.
Liz: Was that the neuromodulation side of Boston Scientifc?
David: Interestingly, it was the vascular side of Boston Scientific that invested.
As I said before, vascular surgeons perform most amputations in the United States and they continue to manage those individuals afterwards.
From their perspective, if those patients remain in the vascular care pathway, physicians can continue treating other conditions those patients may have, like carotid disease, contralateral limb revascularization or aortic disease.
So Boston Scientific’s Vascular Division understood the patient population and the opportunity to help vascular surgeons treat those patients more holistically.
Liz: Talking of investors, congratulations on the recent news. Can you tell us a little bit about it here?
David: Thanks Liz . Thanks. One of our existing investors, Rob Lake, and the team at Trinity Capital, a key international alternative asset manager, have committed $35 million in growth capital as we move into real-world adoption.
This new capital will help us scale the commercialization of the Altius system and expand into new markets.
It will help us on our mission to Relieve Pain and Restore Life for this underserved patient population.
Liz: That is fantastic David. That is truly fantastic. I’m interested – this was a PMA was it. Can you tell me about the regulatory pathway and the current reimbursement situation ?
David: You’re right. From a regulatory standpoint, the Altius System received FDA approval through the PMA pathway, the most rigorous and complex pathway for medical devices.
That approval allows us to bring a dedicated therapy to individuals with chronic post-amputation pain, who historically has had very few treatment options.
On the reimbursement side, we have Category I CPT codes, and the team has had good success getting coverage for patients that show documented medical need.
There is still work to do with some insurers, but we are seeing increasing recognition that this is an important therapy for a population that has been underserved for a long time.
Liz: When people talk about medical technology companies, the conversation often turns to total addressable market. How do you think about the TAM for Neuros?
David: Sadly, it is getting larger. Diabetes and peripheral vascular disease could double the rate of amputations by 2050. That’s why I am watching those markets closely – cheering them on.
We do not need more amputations. We need more people to keep their limbs.
We don’t want a bigger TAM. That is not our goal.
The goal should be preventing amputations in the first place.
We need more people to keep their limbs.
If better diabetes management or better vascular care reduces amputations, that is a good thing.
We are there to pick up afterwards. Our focus is helping the people who are living with amputations.
Liz: When you talk about the TAM like that, it sounds like the mission matters more than the market size.
David: It absolutely does.
What keeps me up at night is not revenue targets or valuation. It is the people living with this pain.
There is roughly a 30% suicide rate in this population.
If someone decides to take their own life because of their pain, that is something I struggle with.
So when I think about success for this company, it is not just about business metrics. It is about whether we have treated as many individuals as possible and given them a real option for relief.
Liz: One theme that often comes up in medical technology is competition. Companies fighting for market share. How do you think about that?
David: I think the industry sometimes approaches that the wrong way. As I said, I am cheering the diabetes and peripheral vascular markets on.
I also respect many of the players in the Neuromodulation space. We have a common goal and often different approaches. No one size fits all. We need ‘different’.
However, when companies in any space start competing by lowering price to gain market share, honestly- all I can say is I think that strategy is one of the dumbest things the industry can do.
When prices drop, reimbursement drops with it.
And when reimbursement drops, companies have less money to invest in research, development, and expanding indications. In the long run that hurts everyone-companies, physicians, investors, individuals needing this therapy.
So, we should be collaborating more, particularly around generating strong clinical evidence and helping payers understand the value of these therapies.
If we do that well, more individuals get treated and the entire field moves forward.

THE PERILS OF SUCCESS
Liz: If we look a few years ahead, what would success look like for you and the company?
David: Success would mean treating as many individuals as possible and establishing this therapy as part of the standard of care.
Liz: One question physicians always ask with neuromodulation therapies is about durability. How long does the therapy continue working for?
David: That is an important question.
Some of our earliest ‘patients’ from the pilot studies are still using the therapy many years later. In fact, our longest ‘patient’ has now been on therapy for sixteen years.
For physicians and individuals, that kind of durability really matters. It tells you the therapy can continue helping people long term. That means so much to us at Neuros.
If every individual with chronic post-amputation pain had access to this therapy, that would be a huge win.
But success also brings another kind of concern. It brings perils.
If a company like Neuros gets acquired, there is always a risk that the mission changes and the technology is not maximized for the individuals it was built for.
We have a duty of responsibility to the individuals using our technology. They have been let down previously.
One of my responsibilities if we ever get to a transition, exit, sale will be to do everything I can to safeguard their quality of life.
That is something I think about a lot.
NEUROTECH: THE FUTURE
Liz: Looking beyond Neuros for a moment, where do you think neuromodulation as a field could have the biggest impact in the future?
David: There are several areas that are exciting.
If we could truly address diabetic neuropathy, that would be huge. Diabetes is driving a lot of the vascular disease and amputations we see today.
Migraine is another area. Those individuals suffer tremendously and the drug regimens are often difficult.
We are also seeing interesting work in stroke recovery and even heart failure using neuromodulation approaches.
So, there is still a lot of opportunity ahead for this field.
There is also the consumer side. You featured Pulsetto last week and Povilas. If individuals are beginning to use this more mainstream, it may lead to greater health awareness, trend detection and maybe earlier interventions – pre-diabetes or picking up quickly on changes in blood pressure.
CLOSING THOUGHTS
Neuros Medical is attempting to establish a new standard of care for a population that has historically had few dedicated treatment options.
Achieving that goal means more than regulatory approval. It will depend on physician adoption, reimbursement alignment, and long-term trust within the amputee community.
The tension at the centre of the story remains unusual.
The company’s commercial opportunity grows if amputations increase. Yet David Veino, its CEO, openly hopes those numbers fall.
In an industry often defined by expansion narratives, David Veino’s focus remains fixed on something far simpler: whether individuals living with chronic post-amputation pain finally have a therapy that can restore their lives.
Do You Want to Be Featured?
If you are a CEO or Co-Founder, and would like to be featured within my Leader In The Spotlight NeuroTech, please reach out to me.
Are you a VC, FO, KOL, researcher, or clinician with a specialist interest in NeuroTech – then please also get in touch, as I want to have a well rounded perspective.
Thank you.
Understanding The Brain- The Ultimate Frontier.
About the author

Liz is our Principal Consultant at Cruxx, covering neurotechnology markets. She is a very well liked, inisghtful, highly connected and respected, champion of start-ups and the people within them. She is unique.
Passionate and energetic, she is driven by finding the best talent today, for the healthcare technology of tomorrow.
She works globally, building impactful teams and headhunting unique talent for start-ups and VCs pushing the frontiers of medical innovation and technology.
If you are growing your team reach out. She specialises in trans-atlantic team builds, USA, Europe and wider.
From R&D, clinical and regulatory to C-suite and Board level, Liz is exceptional and relentless at sourcing visionary, talented individuals for our clients.