LEADER IN THE NEUROTECH SPOTLIGHT: TETIANA ALEKSANDROVA, CO-FOUNDER & CEO, SUBSENSE

IT BEGAN WITH CHEMISTRY AND A CEILING
Liz: Tetiana, let’s start at the beginning. How did you end up in the neurotechnology space? What were you doing before Subsense, and where did your interest in NeuroTech first come from?
Tetiana: My interest in NeuroTech began with Chemistry. As a child, I was fascinated by how things work at the smallest level, how molecules interact, how materials behave, and how small changes can affect an entire system.
I did not formally study Chemistry at university. I chose Economics and later completed an MBA because I was equally interested in understanding systems, markets and how organisations grow. I wanted to build companies.
Neurotechnology felt like the natural convergence of those interests. It combines Material Science, Signal Processing and Human Systems. That intersection is what drew me in.
About seven years ago, I began exploring brain–computer interfaces (BCIs). For me, it felt like the ultimate integration of science and systems thinking. You are working at the level of neurons and signals, but you are also thinking about how humans interact with technology and how intelligence scales. That led to my first company, which focused on non-invasive EEG-based brain–computer interfaces.
Liz: Tell me more about that period. What were you building with EEG, and what did it teach you?
Tetiana: We were building a completely non-invasive BCI based on EEG and machine learning. The goal was to enhance cognitive functions and explore how accurately we could decode brain signals from the surface.
EEG works better than many people think. When we started, some scientists told us it would not work at all. Two years later, those same people were interested in discussing the results.
But I also learned something important. The signal is noisy. You are reading through the skull. For certain applications that is enough. But if you think about the long-term potential of BCIs, you begin to see the limitations.
It showed me both what was possible and where the ceiling was.
Liz: Leaving something that works is never easy. How did you come to the decision to step away and start again?
Tetiana: It wasn’t an impulsive decision. I’m very pragmatic. Before doing anything, I needed to know whether another path was even possible.
So I spent about a year and a half exploring. I spoke with nearly a hundred experts across chemistry, physics, neuroscience and nanoscience. The question was simple: are we breaking any laws of physics? If the answer had been yes, I would have stopped.
What I kept hearing was that it was theoretically possible. The constraints were engineering and biology, not physics.
As I began forming the early team and future collaborators around the idea, that technical validation gave us confidence to move forward.
I realised that if I didn’t pursue it, I would always wonder whether we could have gone further.
It was a calculated decision, not to abandon what worked, but to move beyond what I now understood were structural limits.
NOT BREAKING PHYSICS
Liz: At what point did this shift from exploration to commitment? When did Subsense become a company rather than a research question?
Tetiana: The turning point was when the discussions stopped being theoretical. Once I had spoken to enough experts to understand that we were not breaking physics, the question changed from “is this possible?” to “who is going to build it?”
That is when Subsense began to take shape.
I knew this would require a different kind of company. It would not be software-first. It would require materials science, laboratory capability and hardware integration from the start. So, the next step was assembling the right technical collaborators and building infrastructure around the idea.
At that stage, the risk was no longer scientific impossibility. The risk was execution.
Liz: So what is Subsense addressing and how?
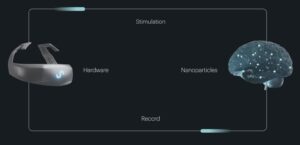
Tetiana: I co-founded Subsense with Artem Sokolov, and we are developing a non-surgical, nanoparticle-powered BCI bio-platform that is bidirectionally designed to precisely read and stimulate neural activity without implanted hardware. We are addressing the gap between high-precision implantable devices and lower-fidelity non-invasive tools, which limit access, scalability, and repeat use in neurological care and beyond.
Liz: You describe it as bidirectional. What does that mean in practical terms?
Tetiana: It means the system can both read neural activity and write back to the brain through stimulation.
Reading allows us to detect and decode neural signals.
Writing allows us to modulate specific circuits in response.
Both functions are necessary to create a true closed-loop interface. Without reading, you do not know what is happening in the brain. Without writing, you cannot influence it. The combination is what makes it an interface rather than just a monitoring tool.
INTRANASAL NANOPARTICLES
Liz: Before we talk about clinical applications, can you explain how this actually works? How do the nanoparticles enter the brain and how are they controlled?
Tetiana: We are exploring two primary delivery approaches. The first, and our preferred path, is intranasal delivery. Nanoparticles can travel along the olfactory nerve from the nasal cavity directly into the brain, which allows us to bypass the blood–brain barrier.
The second approach is systemic delivery through the bloodstream. In that case, nanoparticles are engineered to cross the blood–brain barrier under specific biological conditions.
Once inside the brain, localisation becomes critical. We design the particles so they can be guided and concentrated in specific regions using magnetic fields and surface chemistry. This is not about dispersing particles randomly. It is about precision.
Another key factor is duration. Nanoparticles can be engineered for stability or for controlled degradation. Depending on the application, they can remain active for a defined period or be designed to clear over time. That flexibility is something permanent implanted hardware does not offer.
Liz: And once the particles are in place, how does the read-and-write process function?
Tetiana: We use two types of nanoparticles, each serving a distinct role in the closed-loop system.
For reading neural activity, we use plasmonic nanoparticles. These respond to near-infrared light emitted by an external device worn on the head. When nearby neurons become active, the optical properties of the particles shift, and that change can be detected and decoded.
For stimulation, we use magnetoelectric nanoparticles. When exposed to a magnetic field, they generate a small electrical signal that can stimulate adjacent neurons.
Together, this creates a closed-loop interface. We can detect neural activity, interpret it, and respond with targeted modulation without implanting permanent electrodes.
Liz: Where does this sit compared to invasive and non-invasive systems?
Tetiana: Traditional invasive systems implant electrodes into the brain. That gives high fidelity but requires surgery. Non-invasive systems stay outside the skull but have limited precision.
Our system interacts inside brain tissue because the nanoparticles are close to neurons, but we do not implant hardware. It is invasive in interaction but non-surgical in procedure. It creates a new space between the two.
It is a third way.

Liz: How does this compare to the current standard of care and what assumptions are you challenging?
Tetiana: Current standards rely either on surgically implanted systems or external approaches with limited resolution. We aim to achieve clinical-grade neural access while avoiding permanent implants and surgical risk.
We are challenging the assumption that effective and highly accurate neuromodulation must require permanently implanted hardware. A non-surgical approach may be better aligned with long-term neurological care and broader accessibility.
Liz: You are also working with research institutions. How do those collaborations fit into the development process?
Tetiana: This field is inherently multidisciplinary. No single group has all the expertise required.
We collaborate with researchers at UC Santa Cruz on plasmonic nanoparticle development and with ETH Zurich on magnetoelectric materials. These are key individuals who have spent years studying the underlying physics and chemistry.
Our role is technology transfer. We integrate academic knowledge into a unified platform that can function in a real-world system. Much of what we do is taking validated scientific principles and engineering them into a deployable product.
Liz: With all the research and innovation happening across neuro and adjacent spaces, what conditions do you think are closest to a breakthrough?
Tetiana: We see Parkinson’s disease, epilepsy and motor disorders as the most likely to see breakthroughs. These areas already demonstrate benefit from device-based therapies but face adoption barriers due to surgery.
This is where our technology fits. In the near term, our focus is clearly on neurological diseases where circuit-level modulation has already demonstrated benefit.
In the not too distant future, mid-term horizon, the platform has broader potential. Publicly, we have discussed domains such as sensorimotor recovery of vision, hearing and movement. There is also research interest in inner speech decoding and thought translation, as well as integration with artificial neural networks.
There are also potential applications in mental health regulation, including monitoring neural markers associated with mood, or identifying appetite regulatory mechanisms.
However, the immediate priority is demonstrating safety, reproducibility and translational potential in defined clinical models.
Liz: Building something at this level requires capital discipline. What have you raised so far and how has it been deployed?
Tetiana: We raised an initial $17 million seed round led by Golden Falcon Capital, with participation from private investors, and we have since expanded total financing to $27 million with Golden Falcon Capital. That capital has been directed almost entirely into research and development: nanoparticle sensing, in vitro stimulation experiments, biosafety programs and hardware miniaturisation.
We recently opened our laboratory and engineering facility in Palo Alto to bring more of that work in-house.
Liz: So, what would success look like by the end of 2026?
Tetiana: We are conducting preclinical studies in mouse models and preparing for larger-animal work. Success would mean reliable signal detection, controlled stimulation and safety validation in biological systems.
We have also initiated early engagement with the FDA to understand the regulatory pathway for a non-surgical interface.
Liz: You have done a lot with relatively modest capital given the technological depth. What achievement are you most proud of so far?
Tetiana: Without a doubt, it was our early breakthroughs in materials development and in vitro nanoparticle stimulation and signal reading. Those results validated our scientific direction and led to additional investor support, bringing total financing to $27 million. We are preparing for a broader Series A in 2026 as we move from preclinical validation toward clinical translation.
THE NEXT FRONTIER – TELEPATHY, COGNITIVE AUGMENTATION
Liz: If we project forward twenty years, what could this realistically change?
Tetiana: Over a longer horizon, if we can reliably read and modulate neural circuits without surgery, BCIs could fundamentally change how humans interact with digital systems.
As neural signal fidelity improves, concepts like external memory capacity or cognitive augmentation become technical areas of exploration, thought transmission – telepathy, or aspects of memory preservation become scientific questions rather than purely philosophical ones. Some people describe that as immortality. I would describe it more cautiously as continuity.
However, these are longer-term research domains. They are theoretical extensions of high-fidelity neural access. It could be 20 years from now or more.

Liz: Tetiana, this has been thoroughly enjoyable. You have spoken about collaboration. Building on that idea, what could we achieve if there was broader collaboration across NeuroTech?
Tetiana: We believe closer collaboration across materials science, clinical neuroscience and device development will accelerate progress. Our academic partnerships show how working across multiple disciplines can shorten development cycles and improve decision-making early in development.
At the same time, aligning early on ethics, safety and scalability helps ensure that innovation translates into responsible, real-world impact. That is critical for patient safety and for trust. After all, the reason we are doing this is for patients. They need to be at the centre of everything we do.
Liz: Tetiana, thank you. This has been an insightful and complete eye-opening discussion for me. I can tell you I have thoroughly enjoyed it.
Do You Want to Be Featured?
If you are a CEO or Co-Founder, and would like to be featured within my Leader In The Spotlight NeuroTech, please reach out to me.
Are you a VC, FO, KOL, researcher, or clinician with a specialist interest in NeuroTech, I would also love to feature you.
Thank you – let’s work together to better understand the ultimate frontier – the brain.
About the author

Liz is our Principal Consultant at Cruxx, covering neurotechnology markets. She works globally in the MedTech space, building impactful teams and headhunting unique talent for start-ups pushing the frontiers of medical innovation and technology. From R&D, clinical and regulatory to C-suite and Board level, Liz is exceptional at sourcing visionary, talented individuals for our clients across Europe and the US.

